
This machines are standard pieces of equipment in every chemistry department.Īs we discussed early on about enthalpy phase transition, we will try to discuss briefly for some phase transitions. The power input into the heaters gives the enthalpy of the transition. This measurement uses two heaters and maintains the temperature of a sample and a reference.  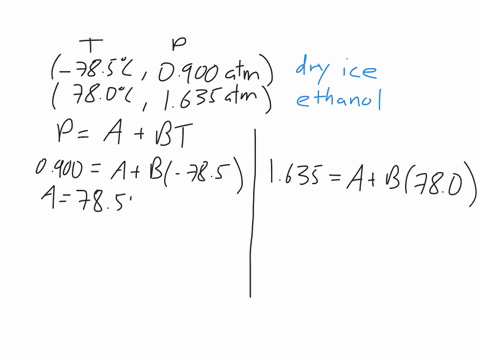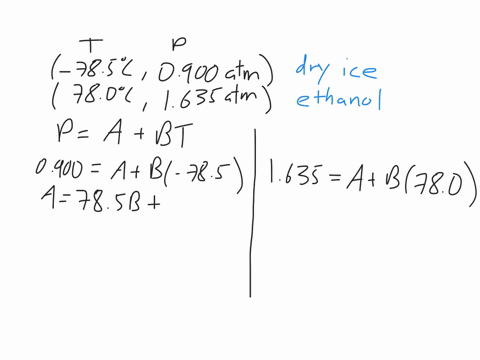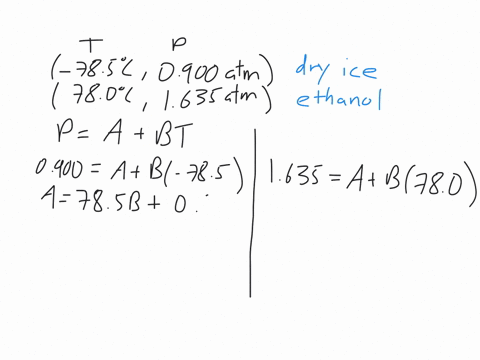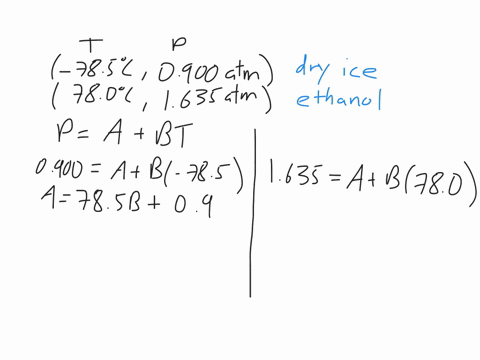
The changes in enthalpy is commonly measured by a differential scanning calorimetry (DSC). The schematic diagram of differential scanning calorimetry (DSC) Therefore, the enthalpy of the water goes up by 25 kJ and we can write as ΔH = +25 kJ, not dH as dH is for a tiny change, ΔH for a large (measurable) change. For example, if we supply 25 kJ of energy to heat an open beaker of water, it happens at constant pressure. Moreover, enthalpy is used extensively in chemistry because most reaction take place under constant pressure, usually atmospheric pressure in open system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
